The shelf life of PPE (Personal Protection Equipment), such as medical face masks and isolation gowns, needs to be guaranteed by the manufacturer. To validate if the performance properties of the packaged PPE after 2 years will remain unchanged, the manufacturer needs to perform an “accelerated ageing” exposure protocol.
The CTT Group is equipped and staffed to help PPE manufacturers to perform accelerated ageing tests, according to test method ASTM F1980, as requested by Health Canada.
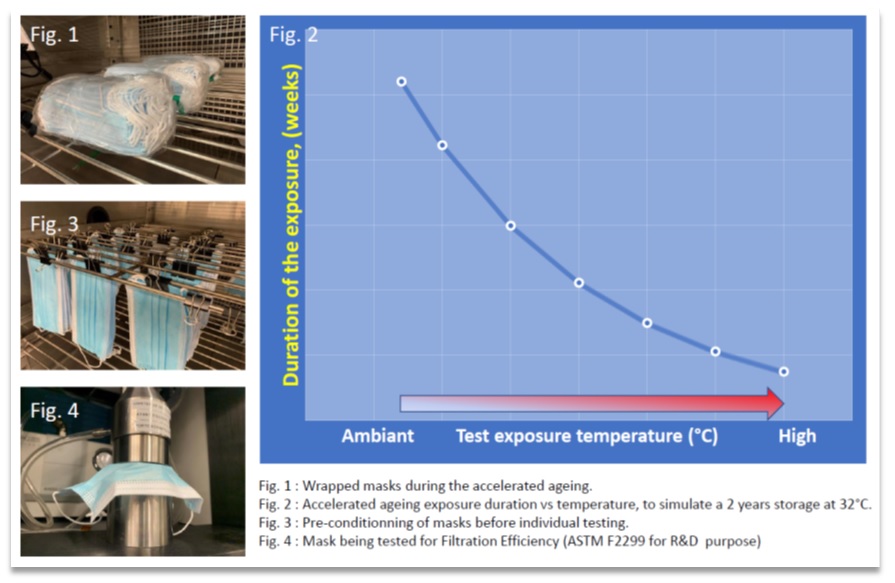
The objective is to predict the real-world ageing of the packaged PPE stored at room temperature for 2 years, by means of a shorter simulated exposure, performed at higher temperatures.
This approach is based on Arrhenius relation describing the rate of degradation of thermoplastic polymers with temperature. Using the oxidation activation energy of common thermoplastics, the rate of reaction doubles approximately with every increase of 10 °C. Thus, the sample will deteriorate about twice as rapidly at 30 °C than at 20 °C, then about twice as fast at 40°C than at 30°C, and so on.
With this knowledge, the ASTM F1980 standard has been established to evaluate the efficiency of medical packaging to storage, with the following limitation: the ageing temperature shall not exceed 60°C.
In the graph below, the duration of accelerated ageing tests (Y-axis) is plotted as a function of exposure temperature (X-axis). This example presents how to determine the duration of the exposure required to simulate 2 years of storage of the packaged PPE at 32°C.
If the storage temperature is expected to be higher, the ageing duration would increase.
Storage conditions should consider manufacturing sites, warehouse, and transport/transits. Moreover, temperature coupled with humidity variations could be used to estimate shelf life of packaged materials.
In a Research and Development process, it is recommended to evaluate the performance properties of your products at the “as received” state, as well as after being subjected to an accelerated ageing protocol.
You may address your questions at: info@gcttg.com



